


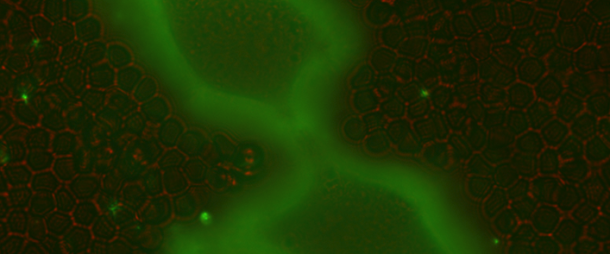

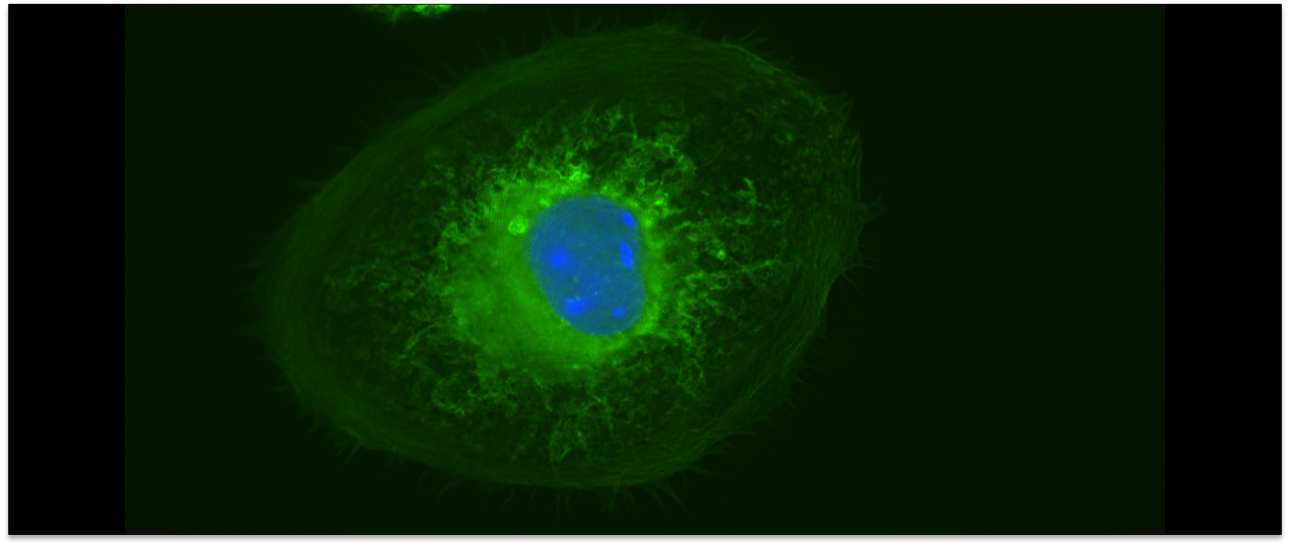
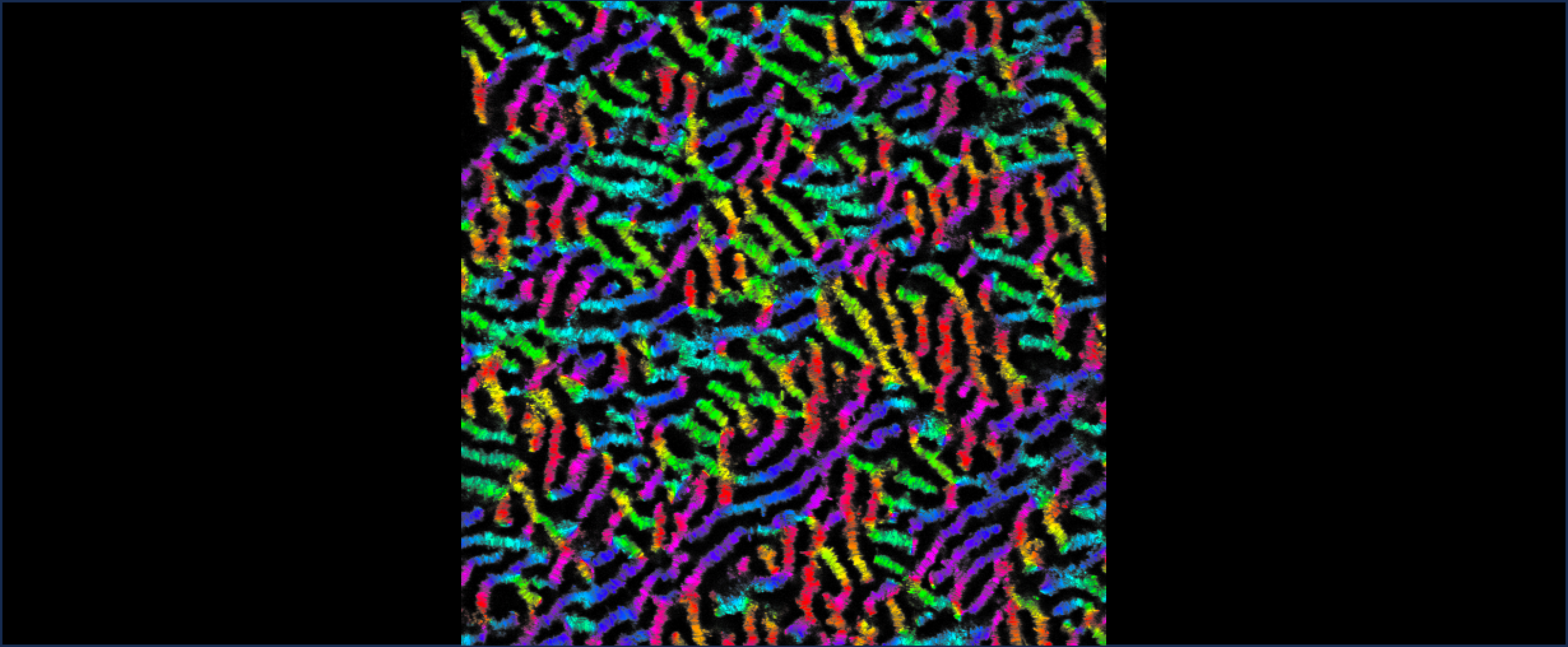

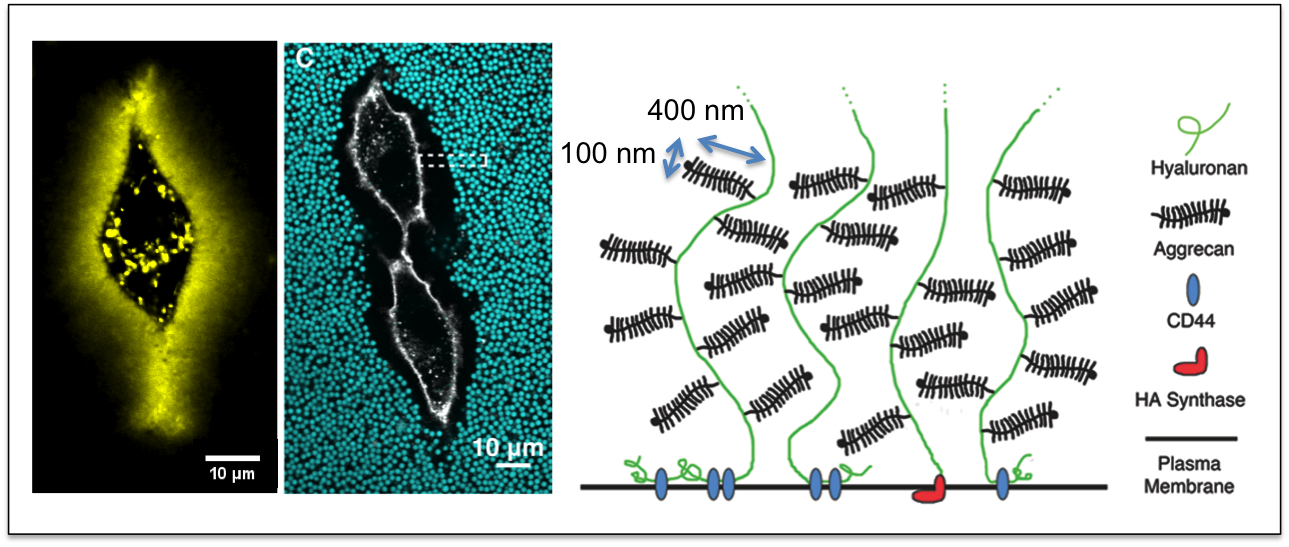

The Curtis lab is focused on the physics of cell-cell and cell-extracellular matrix interactions, in particular within the context of glycobiology and immunobiology. Our newest projects focus on questions of collective and single cell migration in vitro and in vivo; immunophage therapy — an immunoengineering approach — that uses combined defense of immune cells plus viruses (phage) to overcome bacterial infections; and the study of the molecular biophysics and biomaterials applications of the incredible enzyme, hyaluronan synthase.
A few common scientific themes emerge frequently in our projects: biophysics at interfaces, the use of quantitative modeling, collective interactions of cells and/or molecules, cell mechanics, cell motility and adhesion, and in many cases, the role of bulky sugars in facilitating cell integration and rearrangements in tissues.
We value collaborations and hold the philosophy that working closely with biologists, chemists and materials scientists enables meaningful, exciting, high-quality research.

Last updated by JEC on December 3, 2024.